organic compounds now termed as compounds of carbon and organic chemistry defined as the study of carbon compounds Science all organic compounds essentially have carbon the.
Abundance: Carbon is found in every where e.g. plants, animals, coal, petroleum, wood etc. It’s the fourth most abundant element in the universe and fifteenth most abundant element on the earth’s crust.
Atomic number of carbon is 6, electronic configuration is (2, 4), valence is four and it is a non-metal.
Bonding in Carbon: Covalent Bond:
The bond which is formed between two combining atoms by sharing equally one or more pairs of electrons is called a covalent bond or a molecular bond. The compound which is formed using covalent bond is called covalent compound. The molecule which is formed using equally the sharing electrons is called covalent molecule.
There are three different types of covalent bond namely (i) single (ii) double (iii) triple bond.
Single Covalent bond: The bond which is formed by sharing a pair of electrons between the atoms, each atom gives one electron is called single covalent bond.
Hydrogen molecule:
A hydrogen atom has electron in its valence shell. Hence it needs one more electron to attain duplet which is a helium (noble gas) configuration. To meet this need, hydrogen atom shares its electron with another hydrogen atom and forms hydrogen molecules.
Abundance: Carbon is found in every where e.g. plants, animals, coal, petroleum, wood etc. It’s the fourth most abundant element in the universe and fifteenth most abundant element on the earth’s crust.
Atomic number of carbon is 6, electronic configuration is (2, 4), valence is four and it is a non-metal.
Bonding in Carbon: Covalent Bond:
The bond which is formed between two combining atoms by sharing equally one or more pairs of electrons is called a covalent bond or a molecular bond. The compound which is formed using covalent bond is called covalent compound. The molecule which is formed using equally the sharing electrons is called covalent molecule.
There are three different types of covalent bond namely (i) single (ii) double (iii) triple bond.
Single Covalent bond: The bond which is formed by sharing a pair of electrons between the atoms, each atom gives one electron is called single covalent bond.
Hydrogen molecule:
A hydrogen atom has electron in its valence shell. Hence it needs one more electron to attain duplet which is a helium (noble gas) configuration. To meet this need, hydrogen atom shares its electron with another hydrogen atom and forms hydrogen molecules.
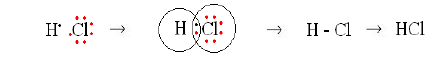
Hydrogen chloride molecule:
Atomic number of hydrogen is 1; hence its valence electron is one. Atomic number of chlorine is 17 and its electronic configuration is (2, 8, 7), hence its has seven valence electrons. By sharing one electron each, hydrogen gets helium structure and chlorine gets argon noble gas structures and hydrogen chloride is formed.
Atomic number of hydrogen is 1; hence its valence electron is one. Atomic number of chlorine is 17 and its electronic configuration is (2, 8, 7), hence its has seven valence electrons. By sharing one electron each, hydrogen gets helium structure and chlorine gets argon noble gas structures and hydrogen chloride is formed.
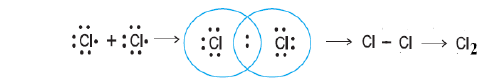
Chlorine:
Atomic number of chlorine is 17 and its electronic configuration is (2,8,7), hence its has seven valence electrons. Two chlorine atoms by sharing one electron each, get argon structure (2,8,8) and forms chlorine molecule.
Atomic number of chlorine is 17 and its electronic configuration is (2,8,7), hence its has seven valence electrons. Two chlorine atoms by sharing one electron each, get argon structure (2,8,8) and forms chlorine molecule.
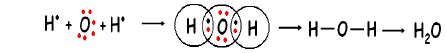
Water Molecule:
Atomic number of hydrogen is 1 and its valence electron is one. Atomic number of oxygen is 8 and its electronic configuration is (2,6) and its valence electron is 6. So, each of the two hydrogen atoms share an electron pair with the oxygen atom such that hydrogen acquires a duplet configuration and oxygen an octet, as a result formed two single covalent bond.
Atomic number of hydrogen is 1 and its valence electron is one. Atomic number of oxygen is 8 and its electronic configuration is (2,6) and its valence electron is 6. So, each of the two hydrogen atoms share an electron pair with the oxygen atom such that hydrogen acquires a duplet configuration and oxygen an octet, as a result formed two single covalent bond.
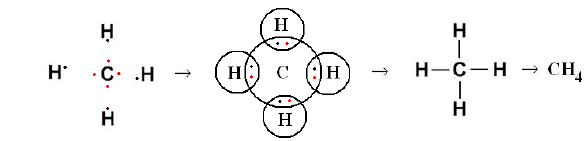
Methane molecule (CH₄):
When methane molecule is formed, one atom of carbon shares four electron pairs, one with each of the four atoms of hydrogen.
When methane molecule is formed, one atom of carbon shares four electron pairs, one with each of the four atoms of hydrogen.
Double Covalent Bond:
When two atoms shares four electrons equally, two electrons from each atoms, forms double covalent bond.
Oxygen Molecule(O₂):
Atomic number of oxygen is 8, electronic configuration (2,6), so it has six valence electrons. So, two oxygen atoms by sharing two electrons each get nearest neon noble gas configuration and forms double bond and forms oxygen molecule.
When two atoms shares four electrons equally, two electrons from each atoms, forms double covalent bond.
Oxygen Molecule(O₂):
Atomic number of oxygen is 8, electronic configuration (2,6), so it has six valence electrons. So, two oxygen atoms by sharing two electrons each get nearest neon noble gas configuration and forms double bond and forms oxygen molecule.
Triple Covalent Bond:
When two atoms share six electrons mutually, forms triple covalent bond.
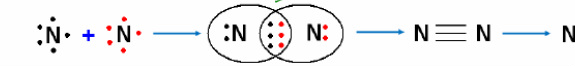
Nitrogen molecule (N₂):
Atomic number of nitrogen is 7, electronic configuration (2,5) and its valence electrons are 5. Two nitrogen atoms, each contributes three electrons and they have three shared pairs electrons between them. Both atoms attain an octet, resulting in the formation of a triple covalent bond.
When two atoms share six electrons mutually, forms triple covalent bond.
Nitrogen molecule (N₂):
Atomic number of nitrogen is 7, electronic configuration (2,5) and its valence electrons are 5. Two nitrogen atoms, each contributes three electrons and they have three shared pairs electrons between them. Both atoms attain an octet, resulting in the formation of a triple covalent bond.
Conditions for the formation of a covalent bond:
(i) Both atoms should have the number of valence electrons four or more.
(ii) Both the atoms should have high electronegativity.
(iii) Both the atoms should have high electron affinity.
(iv) Both the atoms should have high ionization energy.
(v) The electronegativity difference of the two atoms is either zero or negligible.
Properties of Covalent bond:
Intermolecular force is smaller.
Covalent bonds are weaker than ionic bond. Hence melting and boiling point of covalent compounds are less than the ionic compounds.
Science there is no charged particles are present in covalent bond, the covalent compounds are bad conductor of electricity
(i) Both atoms should have the number of valence electrons four or more.
(ii) Both the atoms should have high electronegativity.
(iii) Both the atoms should have high electron affinity.
(iv) Both the atoms should have high ionization energy.
(v) The electronegativity difference of the two atoms is either zero or negligible.
Properties of Covalent bond:
Intermolecular force is smaller.
Covalent bonds are weaker than ionic bond. Hence melting and boiling point of covalent compounds are less than the ionic compounds.
Science there is no charged particles are present in covalent bond, the covalent compounds are bad conductor of electricity

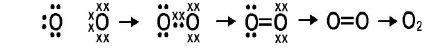
 RSS Feed
RSS Feed